 There is a country song about having too much fun. Its thesis: just like having too much money or a car too fast, there is "no such thing."
There is a country song about having too much fun. Its thesis: just like having too much money or a car too fast, there is "no such thing."
Scientists across the country would add one more item to that list: a research administration office too quick. "Concerns about the amount of time that projects take to move through research administration are not unusual in an institution of this size," said Christopher Brown, Ph.D., vice president for Research. UAB has long been one of the nation’s leading research universities, ranking among the top public institutions for total grants and contracts awarded. And in the past several years, more research projects than ever have flowed to UAB. In 2020, research funding reached a record high of more than $638 million. Since 2016, research awards at UAB have grown 41%.
That does not mean Brown is interested in business as usual. “Time is the coin of the realm,” he said. “That is the measure we use to gauge our efficiency in research administration — are we making things faster and easier for faculty, departmental staff and ourselves?” The entire campus now has a view of some measures of that efficiency on two data dashboards created by the Research Administration Process Improvement and Design (RAPID) Task Force. RAPID launched in fall 2020 with Brown and School of Medicine Dean and Senior Vice President of Medicine Selwyn Vickers, M.D., as executive sponsors. (Learn more about task force membership below.)
Bottom line: more competitive
Except for a niche audience, “administration” and “process improvement” are aggressively uninteresting concepts. Why should anyone else care about the median calendar days needed to redline clinical trial agreements in Q1 2021 or about the median calendar days to complete procedural amendments in 2020, to pull two metrics at random from the Research Business Operations Office and Office of the Institutional Review Board dashboards? The bottom line: These are two markers of UAB’s efficiency in the process of developing cutting-edge treatments for patients across Alabama.
“Scientific merit is the primary driver, but efficient processes make it more likely that UAB will be successful in the extremely competitive world of research funding,” Brown said.
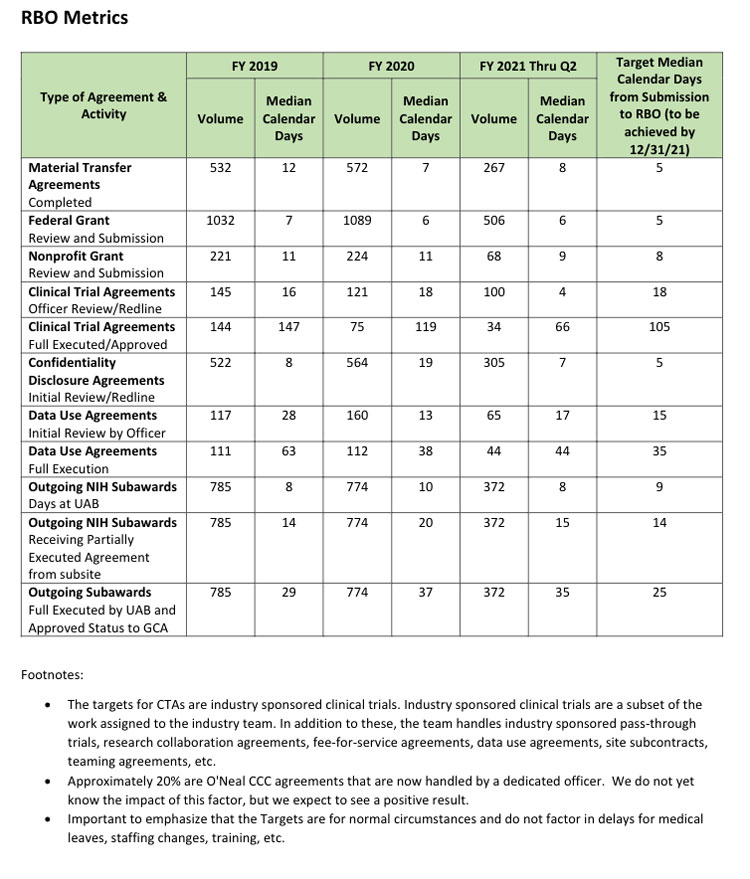 Screenshot of the Research Business Office metrics dashboard. Click to visit on the RAPID site.
Screenshot of the Research Business Office metrics dashboard. Click to visit on the RAPID site.
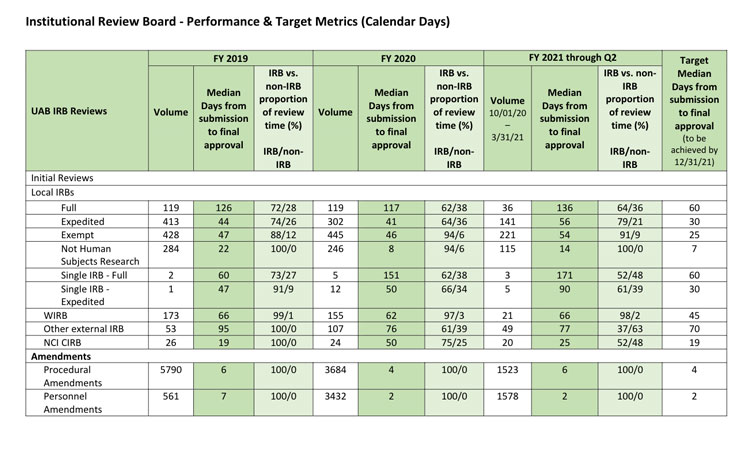 Screenshot of the Institutional Review Board metrics dashboard. Click to visit on the RAPID site.
Screenshot of the Institutional Review Board metrics dashboard. Click to visit on the RAPID site.
Following the data
One particular growth area is in industry-sponsored clinical trials grants, which rose 61% from 2016 to 2019 and are the second-largest research funding source at UAB, after the NIH.
 "Efficient processes make it more likely that UAB will be successful in the extremely competitive world of research funding," said Christopher Brown, Ph.D., vice president for Research.Behind every one of those awards are specialists in compliance, negotiation and the arcane but essential minutiae of federal and state research regulations. Some grants, such as NIH grants, are fairly straightforward — “either accept them or don’t,” said Melinda Cotten, associate vice president for Research Business Operations and director of the Office of Sponsored Programs. Others, particularly industry-sponsored clinical trials, “are an intricate dance” of negotiations about contract language.
"Efficient processes make it more likely that UAB will be successful in the extremely competitive world of research funding," said Christopher Brown, Ph.D., vice president for Research.Behind every one of those awards are specialists in compliance, negotiation and the arcane but essential minutiae of federal and state research regulations. Some grants, such as NIH grants, are fairly straightforward — “either accept them or don’t,” said Melinda Cotten, associate vice president for Research Business Operations and director of the Office of Sponsored Programs. Others, particularly industry-sponsored clinical trials, “are an intricate dance” of negotiations about contract language.
RAPID’s focus is on the number that matters most in the research contract process: time to activation. “Sponsors keep metrics on performance — how fast we turn projects around and how we recruit,” said Jason Nichols, O.D., Ph.D., associate vice president for research engagement and partnerships at UAB and a member of the RAPID Task Force. “From the point we get a call to when we enroll the first patient, it’s the faster the better. We’re often in competitive enrollment scenarios, and we want to see patients as fast as we can so that we can ensure we can bring new therapeutic options to Alabamians.”
The RAPID Task Force's first item of business was to develop a set of metrics tracking historic and projected time to activation across the entire research lifecycle. "One way to improve is to know what the different facets are of time to activation," Brown said. "There are a lot of parallel and sequential processes involved. Once you start publishing how long each step takes for everyone to see, you can find areas to improve and come to agreement on what are reasonable targets to aim for."
"Ultimately, that's what people care about: 'What time can I start?'" said Jonathan Miller, assistant vice president for Research Regulatory Oversight.
Seven real-life examples
Dashboards are all well and good. But you probably want to know how Research Business Operations/OSP and Research Regulatory Oversight/OIRB are speeding up the process.
Cotten and Miller are glad you asked. Results of initiatives emerging from RAPID and the existing research advisory committees already can be tracked on the dashboards. These include metrics for fiscal years 2019 and 2020 and the first quarter of 2021. "Last year was such a strange year that we wanted to make sure we had the 2019 data as a benchmark," Brown said.
1. Breaking through 'intractable contracts'
"We are doing everything we can to make things move faster," Cotten said. OSP recently debuted an electronic tool to accelerate the process of negotiating contracts.
 "We are doing everything we can to make things move faster," said Melinda Cotten, associate vice president for Research Business Operations."There are typical edits we make in contracts," including to the governing law (Alabama is preferred) and to certain make-or-break clauses, Cotten said. "If we are negotiating a clinical trial and the company is not going to reimburse us for subject injury, that's a 'no' deal," Cotten said. "Our new contracting tool gives our OSP officers the ideal language and our least acceptable language for each case, plus columns for every one of the contract language issues that would be a 'go' or 'no go,'" she said. "Negotiating contracts is like a dance back and forth until we get it to the point where we can proceed or have to decline."
"We are doing everything we can to make things move faster," said Melinda Cotten, associate vice president for Research Business Operations."There are typical edits we make in contracts," including to the governing law (Alabama is preferred) and to certain make-or-break clauses, Cotten said. "If we are negotiating a clinical trial and the company is not going to reimburse us for subject injury, that's a 'no' deal," Cotten said. "Our new contracting tool gives our OSP officers the ideal language and our least acceptable language for each case, plus columns for every one of the contract language issues that would be a 'go' or 'no go,'" she said. "Negotiating contracts is like a dance back and forth until we get it to the point where we can proceed or have to decline."
For the thorniest contract decisions, there is a high court: The Research Resolution Council was launched in 2018 with Cotten as chair. Its mission is to act as a final review of contracts that have reached a stalemate — what Cotten calls "intractable contracts." (Council members include Brown and other senior research leaders, including Robert Kimberly, M.D., director of the CCTS, and Barry Sleckman, M.D., director of the O'Neal Comprehensive Cancer Center, as well as Chief University Council John Daniel, J.D., and Brian Burnett, Ph.D., senior vice president for Finance and Administration, and Jason Nichols, Ph.D., associate vice president for Research Engagement and Partnerships.) "So far we've had 49 projects reach the Research Resolution Council at our monthly meetings” during the past three years, Cotten said. "We have said no to 12, two are still pending and we've said yes to 35. The fewer contracts that need to come to the council the better — that means they are moving through our system quicker."
2. IRB + OSP: from linear to parallel
In fiscal year 2020, one important obstacle was removed: the original Policy 005, which mandated that IRB review and approval be complete before industry contracts began routing for signature. "After a thorough peer review, we disconnected the process," so that IRB and OSP reviews run in parallel, Cotten said. "This is how every other university does it."
 "Everything needs to be faster; that's a legitimate concern we all share," said Jonathan Miller, assistant vice president for Research Regulatory Oversight. "Our efforts are focused on unsticking logjams."
"Everything needs to be faster; that's a legitimate concern we all share," said Jonathan Miller, assistant vice president for Research Regulatory Oversight. "Our efforts are focused on unsticking logjams."
3. Faster personnel amendments
That same year, the Research Regulatory Oversight office released an electronic form that split the formerly monolithic process of filing a personnel change on an existing IRB-approved project. "You still have to file an amendment when you make a change, but that previously was the same amendment form, whether you were pulling one person off your study or redoing your whole study — you sat in the same line," Miller said. "We developed an e-form so these two cases could go down separate roads. Now, we get those turned around in a median of two days, whereas at the time the form was introduced that turnaround was about 12 days."
4. Reducing separate data entry
In August 2020, the Office of the Institutional Review Board released the IRB ePortfolio, a suite of dynamic, interactive electronic forms that replaced a system based largely around individual Microsoft Word documents. Text from these documents then had to be re-coded into the IRB systems upon submission. "One of the biggest things about the ePortfolio system is that it eliminated the separate data entry," said Adam McClintock, director of the Office of the Institutional Review Board. "That allowed us to move two full-time employees from data-entry roles to protocol analyst positions through reclassification and retraining."
5. Dedicated personnel make a difference
Speed doesn't just matter to pharmaceutical companies. Major funders such as the National Cancer Institute (NCI) pay close attention to the number of patients enrolling in clinical trials at centers such as UAB's O'Neal Comprehensive Cancer Center. This is because patients' ability to access new and potentially groundbreaking treatments through clinical trials is a key reason for the NCI's Comprehensive designation — and a major factor in grant renewal.
 “Sponsors keep metrics on performance — how fast we turn projects around and how we recruit,” said Jason Nichols, O.D., Ph.D., associate vice president for research engagement and partnerships at UAB and a member of the RAPID Task Force. “From the point we get a call to when we enroll the first patient, it’s the faster the better."The Cancer Center accounts for a large proportion of clinical trials at UAB. So this past year, in another RAPID collaboration, the center funded the hiring of a dedicated OSP officer to work on cancer-related contracts. "It's all about getting the patients in studies faster," said Cotten — not just for the Cancer Center, but all of UAB. "By adding a new OSP officer who just works with Cancer Center trials, our other OSP officers are now able to focus more time on the rest of campus,” Cotten said. “That has been working very well."
“Sponsors keep metrics on performance — how fast we turn projects around and how we recruit,” said Jason Nichols, O.D., Ph.D., associate vice president for research engagement and partnerships at UAB and a member of the RAPID Task Force. “From the point we get a call to when we enroll the first patient, it’s the faster the better."The Cancer Center accounts for a large proportion of clinical trials at UAB. So this past year, in another RAPID collaboration, the center funded the hiring of a dedicated OSP officer to work on cancer-related contracts. "It's all about getting the patients in studies faster," said Cotten — not just for the Cancer Center, but all of UAB. "By adding a new OSP officer who just works with Cancer Center trials, our other OSP officers are now able to focus more time on the rest of campus,” Cotten said. “That has been working very well."
6. ‘Unsticking logjams’
"Everything needs to be faster; that's a legitimate concern we all share," said Miller. "Our efforts are focused on unsticking logjams." That is often through process-oriented work that McClintock excels in, Miller said.
This spring, McClintock worked with the Research Technology Communications team to develop reportable data to track IRB submissions that had received an initial response from the office and were on hold pending more information from investigators. "Prior to this, we didn't have any way to see what was out there waiting on response, so we couldn't take action or send reminders," McClintock said. "When we first got the data and started looking at them there were several hundred initial submissions to the IRB waiting on responses."
Some of these projects had simply been abandoned by investigators, but not all. Sometimes investigators thought they had sent responses but had missed the final “Submit” button. From that initial list of several hundred outstanding initial responses, "over the past few months we have gotten that down to the 30s or 40s," McClintock said. "When we started looking at the median number of days waiting for investigator response it was around 90 and now it is down to two weeks or less."
 "Quite honestly, I would rather have the greater percentage of time with IRB rather than with investigators," said Adam McClintock, director of the Office of the Institutional Review Board. "That means it's in our control to improve it."Unsticking that logjam temporarily turned into a negative as "an onslaught of reviews hit IRB," McClintock said. The number of backlogged submissions temporarily jumped as initial responses that had lain dormant were suddenly reactivated, but this was quickly reduced. The backlog of initial reviews awaiting a first pre-review ballooned (by 78%) and peaked in mid-May. By mid-June, the overall number of submissions awaiting final approval dropped by 35% from the baseline in early March.
"Quite honestly, I would rather have the greater percentage of time with IRB rather than with investigators," said Adam McClintock, director of the Office of the Institutional Review Board. "That means it's in our control to improve it."Unsticking that logjam temporarily turned into a negative as "an onslaught of reviews hit IRB," McClintock said. The number of backlogged submissions temporarily jumped as initial responses that had lain dormant were suddenly reactivated, but this was quickly reduced. The backlog of initial reviews awaiting a first pre-review ballooned (by 78%) and peaked in mid-May. By mid-June, the overall number of submissions awaiting final approval dropped by 35% from the baseline in early March.
7. Workload balancing removes bottlenecks
Going forward, access to timely process data has other benefits. "The same data let us look at workload balancing," McClintock said. "The exempt timeline was taking way too long and 98% of that time was in the IRB office," Miller added. "It was clear the problem was here, so the question was, what can we do about it?" Investigation revealed a workflow bottleneck where final check for several processes was funneling through a single person. So McClintock initiated cross-training to allow three additional staff members to function in that role. "Quite honestly, I would rather have the greater percentage of time with IRB rather than with investigators," McClintock said. "That means it's in our control to improve it."